- Objects can be described based on their properties such as size, shape, texture, and colour.
- The perception of an object is a result of the brain’s interpretation of sensory information received from the physical entity.
- All objects are composed of atoms or molecules, which are the building blocks of matter.
- The properties of the atoms and molecules that make up an object determine its physical and chemical properties.
Is a rainbow an object?
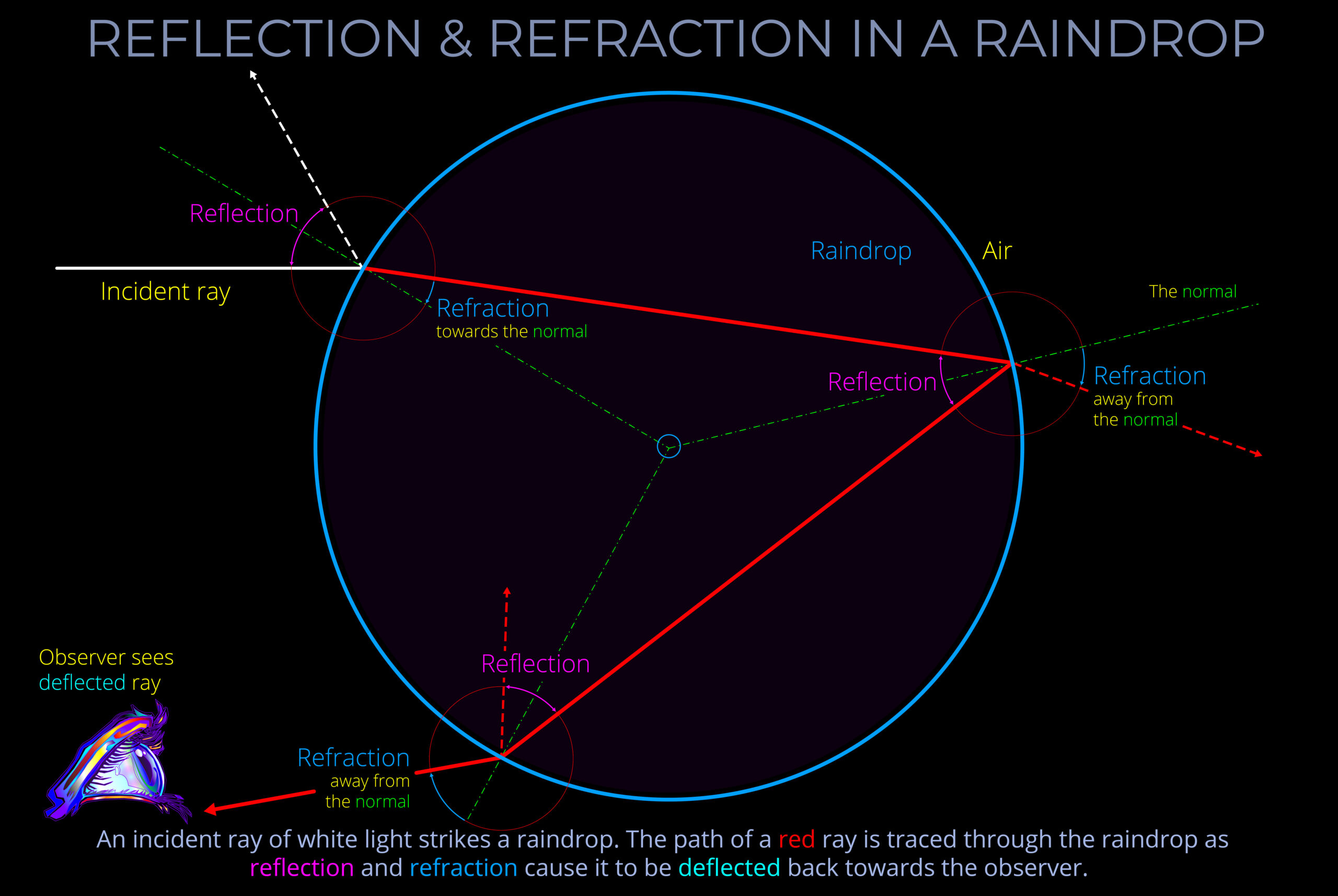
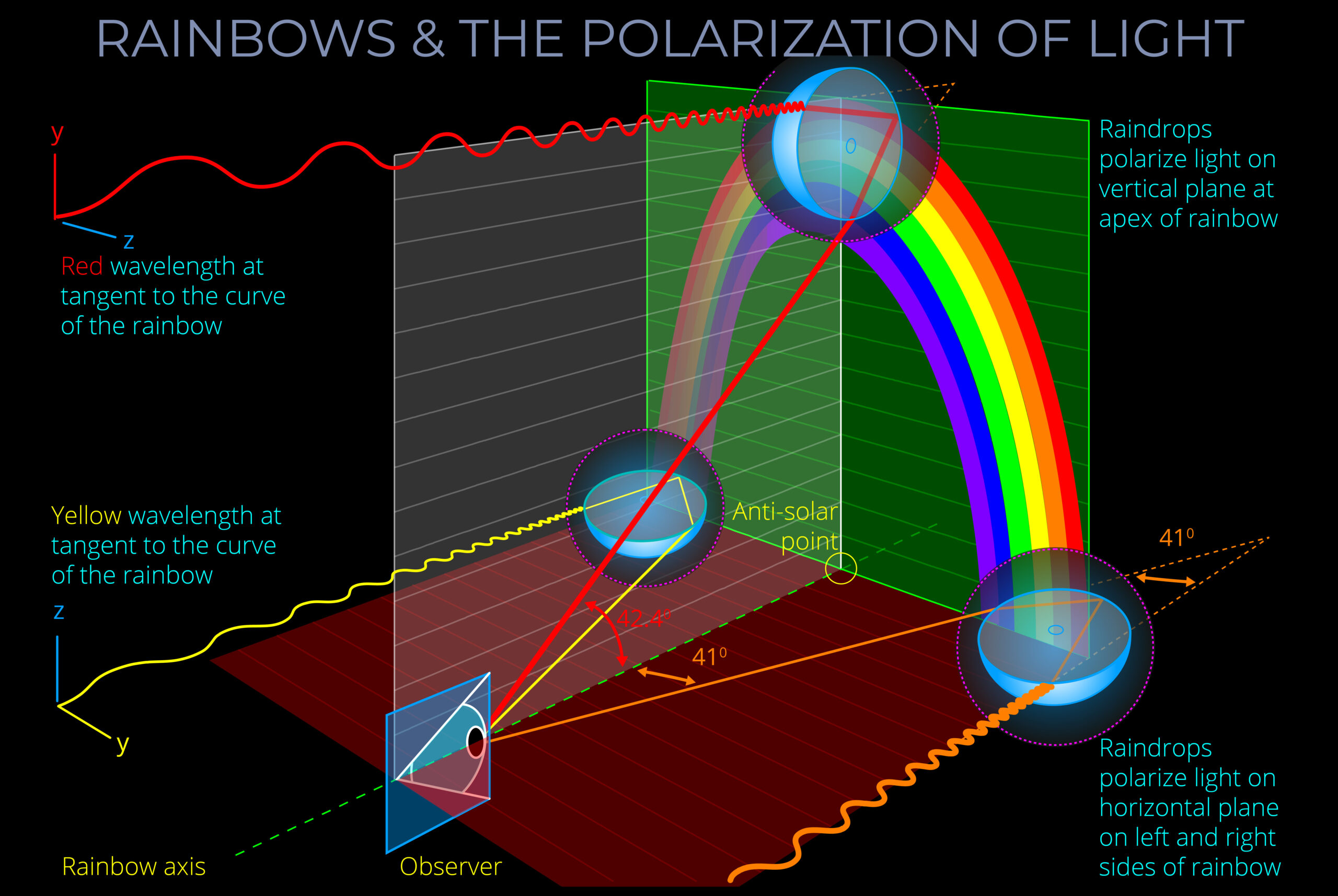
- An atmospheric rainbow is not a physical object but an optical illusion caused by the refraction, reflection, and dispersion of light.
- An atmospheric rainbow is created when sunlight is refracted, reflected and dispersed through water droplets in the air, forming a circular arc of colours in the sky that can be seen by an observer.
- A rainbow is an optical effect, a trick of the light.
About the colour of objects
- Objects are composed of atoms, which bond together to form molecules, elements, and compounds. These different combinations of atoms give objects their unique properties and determine how they react to light.
- Objects appear to have different colours because they absorb some wavelengths of light and reflect or transmit others. The wavelengths that are reflected determine the colour of an object seen by an observer.
- In physics, the absorption and reflection of light are explained as follows:
-
- Atoms are composed of protons, neutrons and electrons. Protons and neutrons form the nucleus and electrons orbit the nucleus.
- The orbit, or energy level, of an electron in an atom can change when the electron gains or loses energy.
- When an electron absorbs a photon of light, it gains energy and moves to a higher energy level so a higher orbit.
- The difference between the initial and final energy levels of the electron is equal to the energy of the absorbed photon.
- When an electron moves to a higher energy level it is in an unstable state and eventually returns to its original lower energy level.
- When the electron returns to its original lower energy level, it emits a photon of light with a frequency and wavelength corresponding to the energy difference between the two levels.
- The difference between any two energy levels of an electron is specific to the type of atom and can be thought of as being equal to a “quantum” of energy, where a quantum is understood to mean an indivisible unit of energy.
- Every type of atom has a unique set of energy levels, and so it emits or absorbs photons of light at specific wavelengths or colours.
- The colour of objects perceived by an observer can be affected by the lighting conditions in which they are viewed and so by the spectral power distribution of the light source.
- The surface texture of objects affects how light interacts with them. Smooth and polished surfaces reflect light in a regular pattern, while rough and textured surfaces scatter light and colour in many directions.
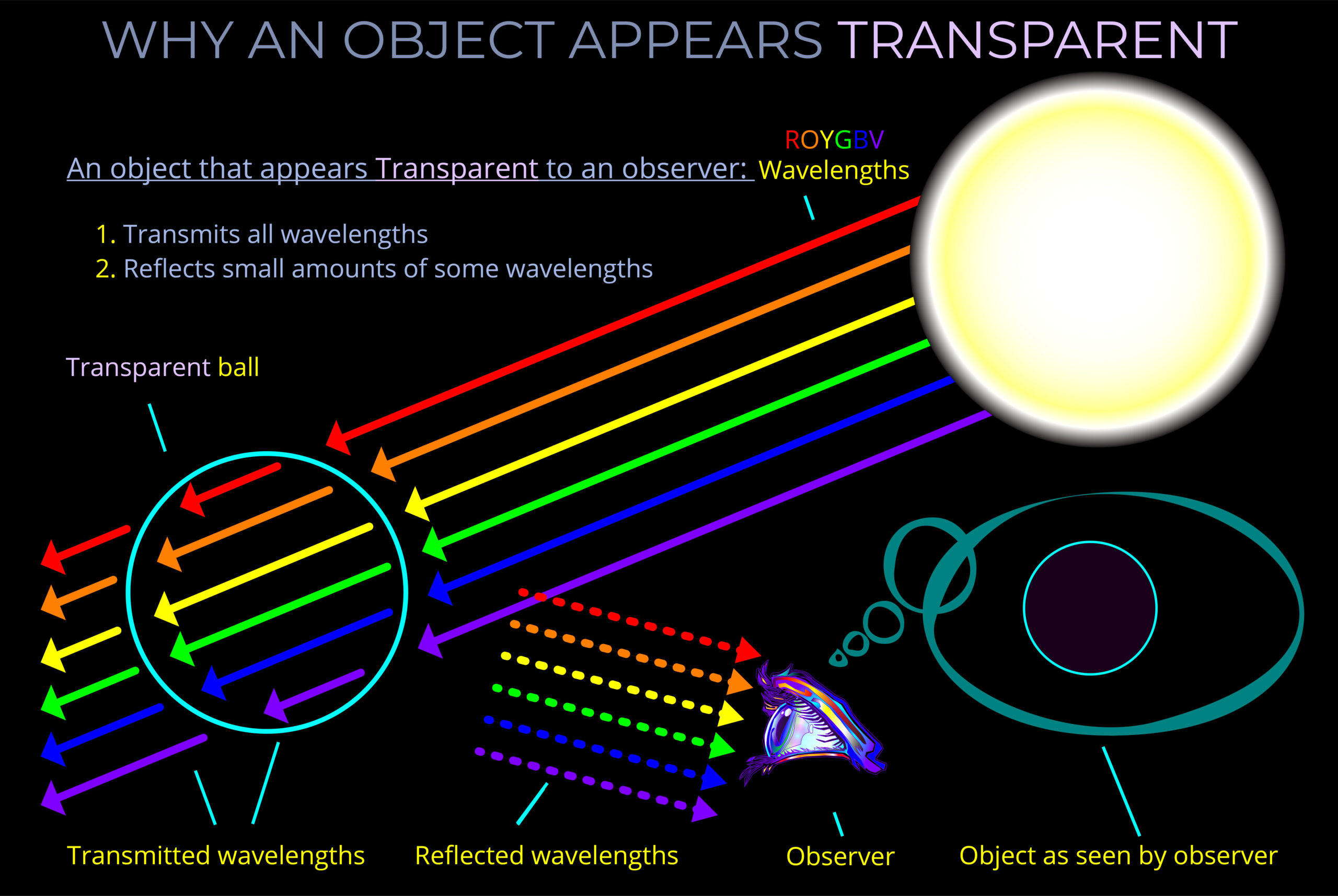
- Transparent objects allow much of the light that strikes them to pass through. The colour seen by an observer is affected by impurities or defects in the material and by the colour of the background against which they are viewed.
- In the case of opaque objects, the surface of the object reflects, absorbs or scatters light, which determines what is seen by the observer.
-
- An object is a physical entity and so a thing that has mass and occupies space.
- Objects can be described based on their properties such as size, shape, texture, and colour.
- The perception of an object is a result of the brain’s interpretation of sensory information received from the physical entity.
- All objects are composed of atoms or molecules, which are the building blocks of matter.
- The properties of the atoms and molecules that make up an object determine its physical and chemical properties.