Electromagnetic Spectrum
£0.00
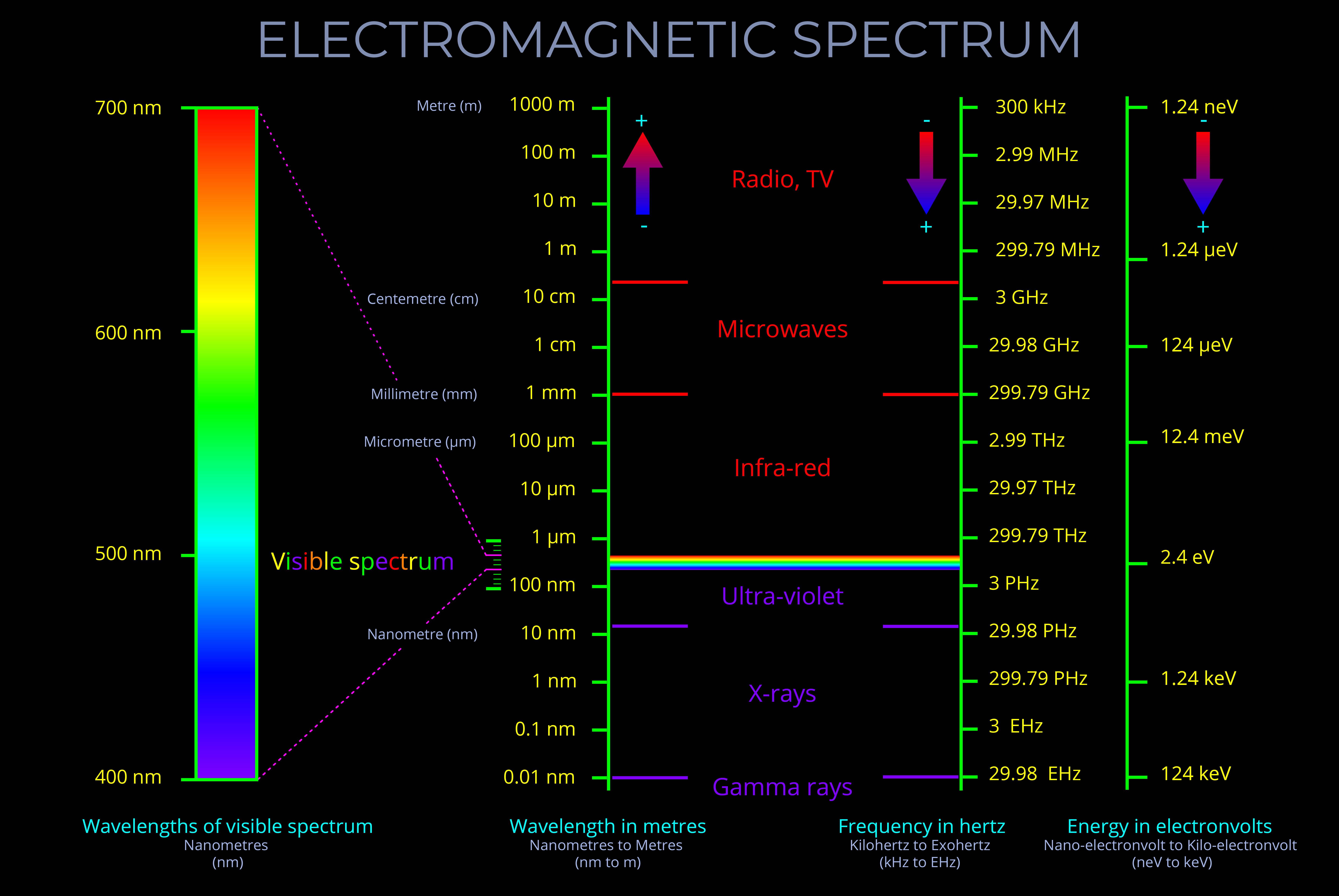
This diagram shows that the electromagnetic spectrum includes waves with all possible wavelengths, ranging from low energy radio waves through visible light to high energy gamma rays.
- Notice that a wave with a longer wavelength has a lower frequency and carries less energy.
- Notice that a wave with a shorter wavelength has a higher frequency and carries more energy.
Remember that:
- The electromagnetic spectrum includes, in order of increasing frequency and decreasing wavelength: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.
- There are no precisely defined boundaries between the bands of electromagnetic radiation in the electromagnetic spectrum. Each band is associated with different properties and applications – think of radios, microwaves, x-ray machines etc.
- Visible light is only a very small part of the electromagnetic spectrum.
- Energy (electromagnetic energy) is the radiant energy (light) transported by electromagnetic waves.
Description
Electromagnetic spectrum
TRY SOME QUICK QUESTIONS AND ANSWERS TO GET STARTED
About the diagram
About the diagram
- This diagram shows that the electromagnetic spectrum includes waves with all possible wavelengths, ranging from low energy radio waves through visible light to high energy gamma rays.
- Notice that a wave with a longer wavelength has a lower frequency and carries less energy.
- Notice that a wave with a shorter wavelength has a higher frequency and carries more energy.
Remember that:
- The electromagnetic spectrum includes, in order of increasing frequency and decreasing wavelength: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.
- There are no precisely defined boundaries between the bands of electromagnetic radiation in the electromagnetic spectrum. Each band is associated with different properties and applications – think of radios, microwaves, x-ray machines etc.
- Visible light is only a very small part of the electromagnetic spectrum.
- Energy (electromagnetic energy) is the radiant energy (light) transported by electromagnetic waves.
- The term light can be used in three different ways:
- Light can be used to mean the whole of the electromagnetic spectrum from radio waves, through visible light to gamma rays. A better term is radiant energy or photon energy.
- Light can be used to mean the range of wavelengths and frequencies that can be detected by the human eye. A better term is visible light which refers to the wavelengths that correspond with the colours between red and violet, the visible spectrum.
- Light can be used to mean the range of wavelengths and frequencies between infra-red and ultra-violet. This usage is useful because the outer limits of the visible spectrum change under different lighting conditions and for different individuals.
- Referring to visible light simply as light is short-hand.
- Visible light is not the same for all living things.
Some key terms
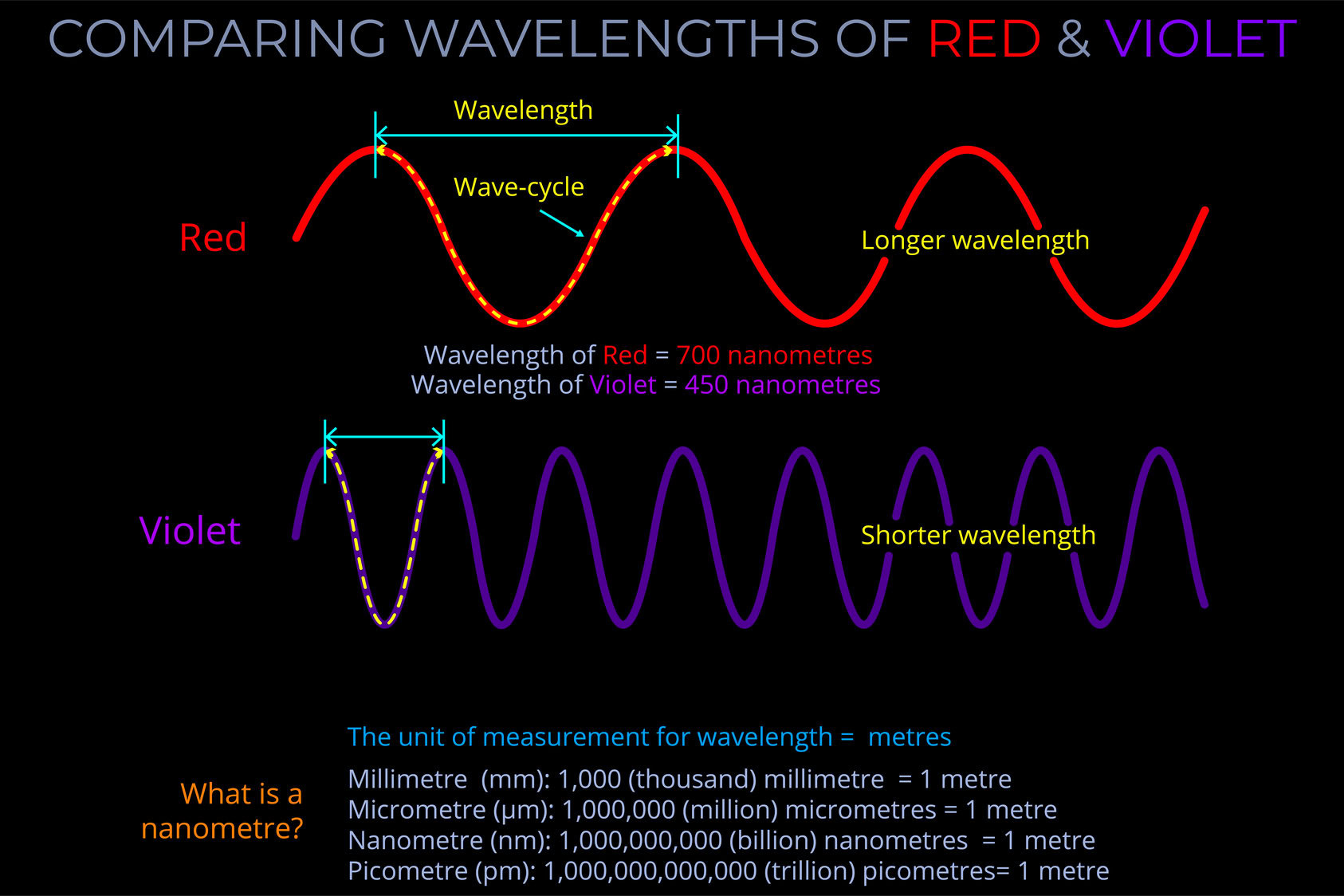
A nanometre (nm) is a unit of length in the metric system, equal to one billionth of a metre (1 nm = 1 × 10⁻⁹ metres). It is commonly used to measure extremely small distances, particularly at the atomic and molecular scale.
- In the context of light and electromagnetic radiation, a nanometre is often used to describe wavelengths of visible light.
The wavelength of visible light ranges from about 700 nm (red) to 400 nm (violet). - Nanometres are also used to measure components like the thickness of materials, the size of particles in nanotechnology, and the spacing between atoms in a crystal lattice.
Energy is a property of matter and fields, which can be transferred between systems or transformed into different forms but cannot be created or destroyed.
- Everything contains energy including all forms of matter and so all objects.
- Energy is evident in all forms of movement, interactions between, and changes to the forms and properties of matter.
- At an atomic level, energy is evident in the movement of electrons around the nucleus of an atom. Energy is stored in the nucleus of atoms as a result of the forces that bind protons and neutrons together.
- Energy can be transferred between objects, and converted from one form to another, but cannot be created or destroyed.
- Everything in the universe uses energy in one form or another.
- When it comes down to it, matter is energy.
- Light has energy but no mass so does not occupy space and has no volume.
- Energy is often described as either being potential energy or kinetic energy.
- Energy is measured in joules.
An electronvolt (eV) is a unit of energy commonly used in atomic, nuclear, and particle physics to measure the energy carried by individual particles and electromagnetic radiation. It’s a convenient unit because the energies involved in these fields are much smaller than those we encounter in everyday life.
- Electronvolts can be used to measure the energy of elementary particles, including photons, which are the smallest units of electromagnetic radiation (quanta of the electromagnetic field).
- One electronvolt is equal to the energy gained by a single electron when it is accelerated across a potential difference of 1 volt.
- Photons (quanta of light) travelling through this same potential difference would also gain 1 eV of energy.
- The electronvolt is not part of the SI unit system. The SI unit for energy is the joule (J). However, joules are too large for many particle-level interactions.
- 1 electronvolt (eV) is equivalent to approximately 1.602 x 10^-19 joules (J).
Wavelength measures a complete wave cycle, which is the distance from any point on a wave to the corresponding point on the next wave.
- While wavelength can be measured from any point on a wave, it is often simplest to measure from the peak of one wave to the peak of the next or from the bottom of one trough to the bottom of the next, ensuring the measurement covers the whole of the cycle.
- The wavelength of an electromagnetic wave is usually given in metres.
- The wavelength of visible light is typically measured in nanometres, with 1,000,000,000 nanometres making up a metre.
- Radio waves, visible light, and gamma waves for example, each have different ranges of wavelengths within the electromagnetic spectrum.
The hertz (symbol: Hz) is a unit used to measure the frequency of electromagnetic waves. It represents the number of wave-cycles per second.
- One hertz is defined as one cycle per second.
- Hertz measure the number of oscillations of the perpendicular electric and magnetic fields in electromagnetic radiation per second.
- Frequency conversions:
- 1 Hertz (Hz) = 1 cycle per second
- 1 Kilohertz (kHz) = 1,000 (thousand) cycles per second
- 1 Megahertz (MHz) = 1,000,000 (million) cycles per second
- 1 Gigahertz (GHz) = 1,000,000,000 (billion) cycles per second
- 1 Terahertz (THz) = 1,000,000,000,000 (trillion )cycles per second
The frequency of electromagnetic radiation (light) refers to the number of wave-cycles of an electromagnetic wave that pass a given point in a given amount of time.
- Frequency is measured in Hertz (Hz) and signifies the number of wave-cycles per second. Sub-units of Hertz enable measurements involving a higher count of wave-cycles within a single second.
- The frequency of electromagnetic radiation spans a broad range, from radio waves with low frequencies to gamma rays with high frequencies.
- The wavelength and frequency of light are closely linked. Specifically, as the wavelength becomes shorter, the frequency increases correspondingly.
- It is important not to confuse the frequency of a wave with the speed at which the wave travels or the distance it covers.
- The energy carried by a light wave intensifies as its oscillations increase in number and its wavelength shortens.