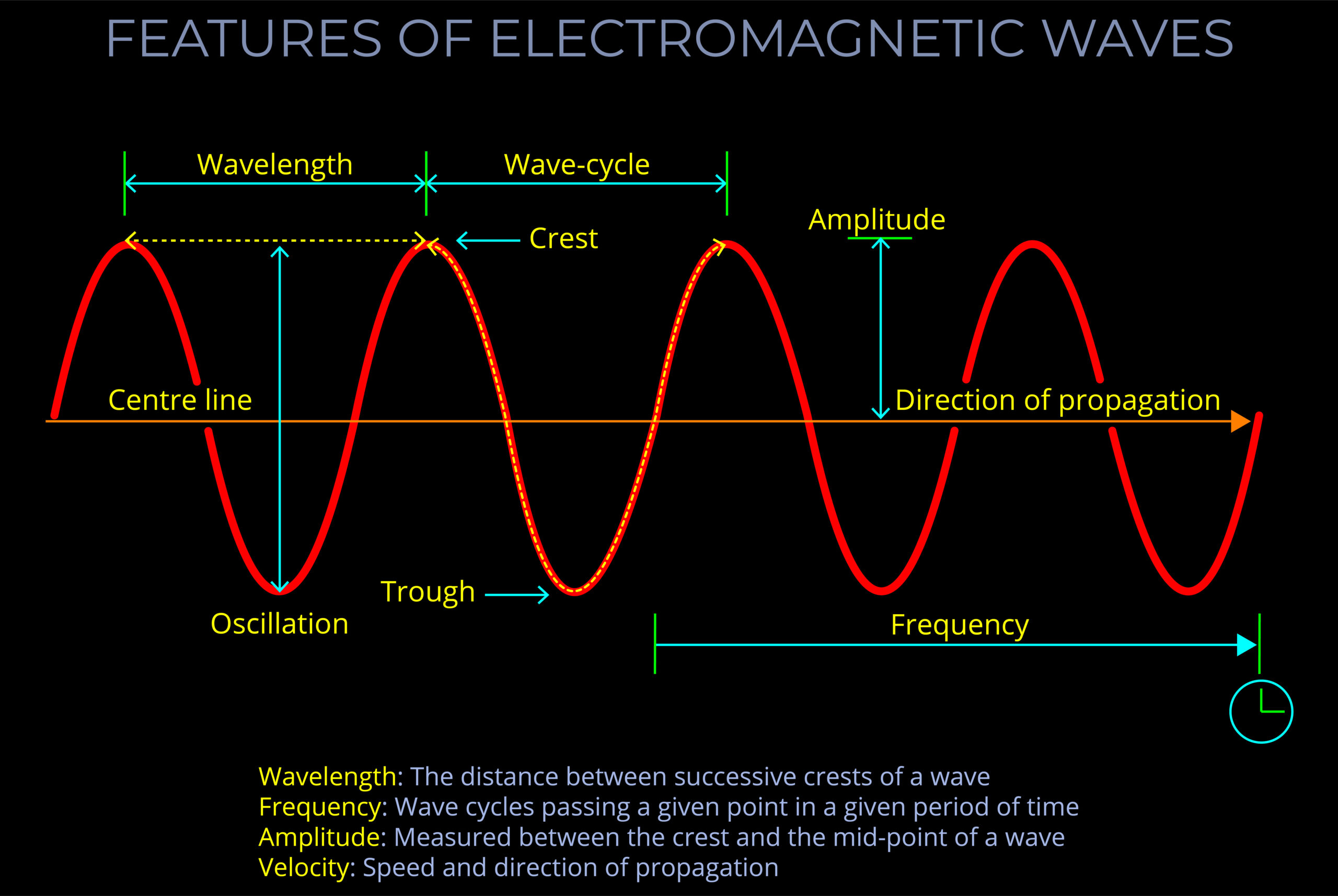
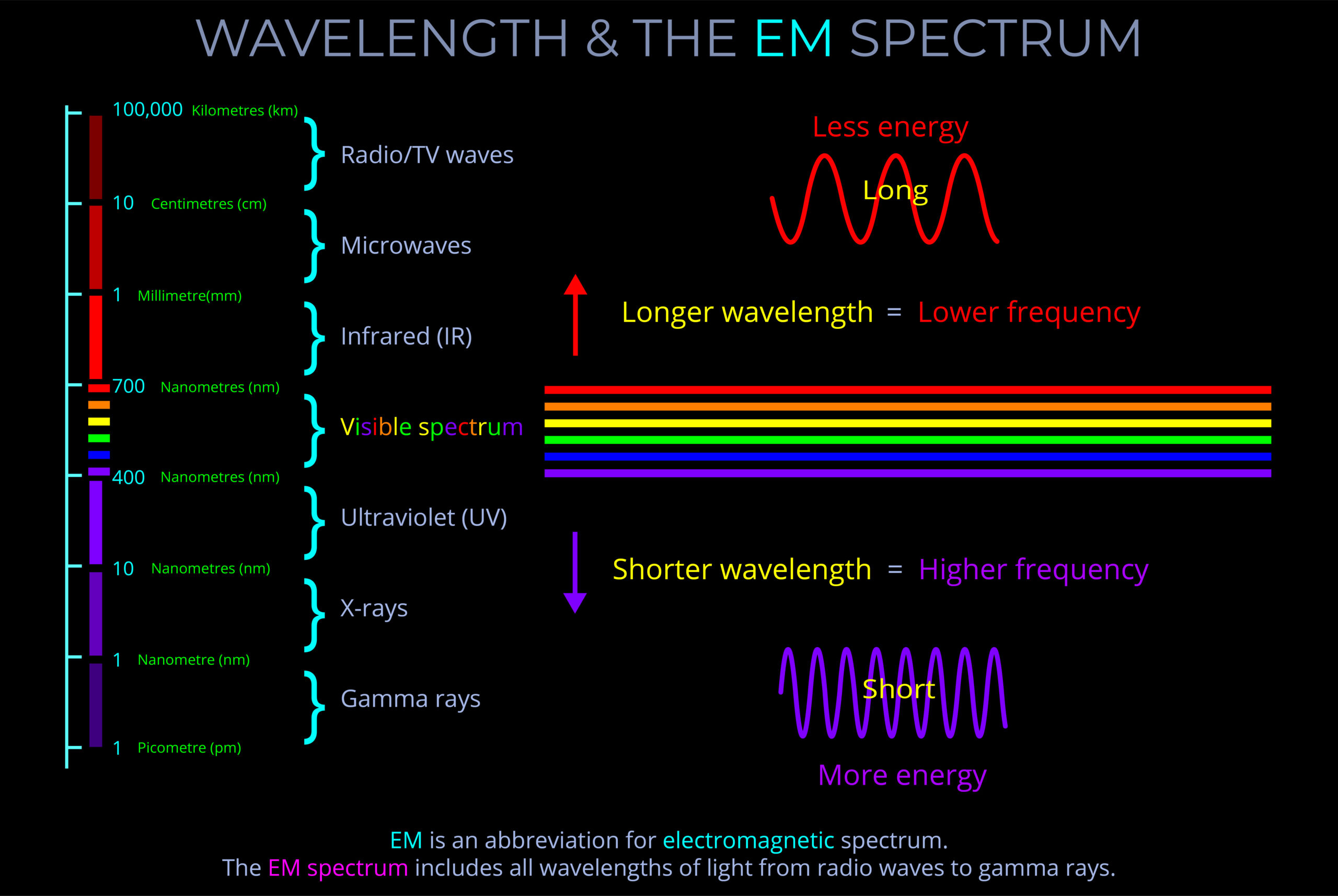
Electromagnetic energy refers to the energy transported by electromagnetic waves.
- The amount of energy carried by an electromagnetic wave is directly proportional to its frequency and inversely proportional to its wavelength.
- Radio waves have the longest wavelength and lowest frequency so carry the least energy among electromagnetic waves, while gamma rays have the shortest wavelength and highest frequency in the electromagnetic spectrum and carry the most energy.
- Electromagnetic waves are composed of oscillating electric and magnetic fields that travel through space at the speed of light.
Photons and electromagnetic energy
- Photons are discrete packets of energy that make up electromagnetic waves.
- The energy of each photon is directly proportional to its frequency and inversely proportional to its wavelength.
- The energy of electromagnetic radiation can be calculated as the total number of photons multiplied by the energy of each photon.
Electromagnetic waves and electromagnetic energy
- Electromagnetic waves can be described in terms of their electric and magnetic fields.
- The electric and magnetic fields of the wave do not carry energy in the same way that photons do.
- Electric and magnetic fields are a manifestation of the energy carried by the wave.
- In other words, the energy of the wave is distributed between the electric and magnetic fields, which are intimately linked and cannot be separated from each other.
- The electric and magnetic fields of the wave do not exist independently of the wave, but rather they are a fundamental part of the wave itself.
Measuring electromagnetic energy
- The energy of electromagnetic waves can be measured using various techniques, depending on the frequency range of the wave being studied.
- In the radio frequency range, measurement requires antennas and receivers capable of converting the electrical energy of the wave into a measurable signal, such as a voltage or a current.
- In the optical and ultraviolet frequency ranges, measurement requires detectors such as photo-diodes or photomultiplier tubes, which convert the energy of individual photons into a measurable electrical signal.
- In the X-ray and gamma-ray frequency ranges, measurement requires specialized detectors such as scintillation counters or Geiger-Müller tubes, which are capable of detecting individual high-energy photons.
- The energy of an individual photon can be calculated using the Planck-Einstein relation, which relates the energy of a photon to its frequency or wavelength. This means that the energy of an electromagnetic wave can be calculated as the product of the number of photons and the energy of each photon.
- In many practical applications, it is more convenient to measure the power (or intensity) of the electromagnetic wave rather than its energy.
- Power is measured in units of watts, and intensity is measured in units of watts per square meter.